Mice injected with a protein containing a mutation found only in the Icelandic genome have shown resistance to Alzheimer’s disease. This mutation reduces the production of proteins responsible for the disease and protects neuronal connections. This opens up new therapeutic avenues in the long term.
Could we one day protect ourselves from Alzheimer’s? A glimmer of hope may arise thanks to a French study by the CNRS and the University of Grenoble Alpes conducted on laboratory mice. These findings were published on June 14, 2024, in the journal Molecular Psychiatry. Neurobiologists inoculated a protein present in a mutated form in the genome of Icelanders, who are particularly resistant to cognitive decline. They observed that, after just one injection, the treated mice did not develop any symptoms of Alzheimer’s disease.
Alzheimer’s disease results from brain degeneration caused by the abnormal accumulation of beta-amyloid (or β-amyloid) and Tau proteins in the brain. However, in recent years, the scientific community has theorized that the toxicity of β-amyloid proteins at synapses – the areas where connections between neurons are established – could be responsible for the neurofibrillary degeneration associated with Tau protein dysfunction. This is why the French researchers focused mainly on β-amyloid proteins, specifically pseudo-prion types.
A prion-like disease in Alzheimer’s
A prion protein is a protein that, if its conformation changes, can become pathogenic and transmit this characteristic to surrounding normal proteins. This is the case, for example, in Creutzfeldt-Jakob disease. «Previously, it was not thought that degenerative diseases could be prion-like. But for about ten years, we have known that the proteins involved in Alzheimer’s disease behave like prions inside the body. This is why we talk about pseudo-prions, especially since they are much less transmissible than prions,» explains Marc Dhenain, a researcher at the Laboratory of Neurodegenerative Diseases (CNRS, CEA, University of Paris-Saclay), and a co-lead author of the study.
However, neurodegenerative diseases are in no way transmitted between individuals, but the inoculation of a pathogenic pseudo-prion in the laboratory can indeed lead to the onset of Alzheimer’s disease. Based on this, Marc Dhenain and his colleagues sought a way to slow down the action of these pseudo-prions.
An «Icelandic mutation» prevents the onset of the disease
Their research led them to Iceland. The small population of the island – around 300,000 inhabitants – allowed for whole-genome sequencing of the country. This census revealed a mutation on a precursor of β-amyloid proteins. By following the cognitive level of these individuals, researchers found that they were protected against Alzheimer’s disease. How is such protection possible?
One hypothesis is that the mutation observed in the genome of Icelanders slows down the uncontrolled production of β-amyloid proteins. Building on this conjecture, French neurobiologists inoculated this mutation into mice modified to develop Alzheimer’s disease and found that those who received an injection retained their memory in the following months.
The scientists assessed the memory of the mice using the Morris water maze experiment: «This is a system where animals are placed in a pool and must find their way to a platform. When memory weakened, the mice could not find their way, while the treated mice still remembered it four months later,» summarizes Marc Dhenain.
Slices of brains that have been colored to detect Alzheimer’s disease lesions.
Protecting synapses by reducing toxicity
But that’s not all. With his team, he introduced more or less toxic forms of amyloids into the brains of mice. The result: the mutation also appears to reduce damage to synapses. «What is really incredible is that we are not used to a single injection still yielding results after four months. This is due to the fact that there is an amplification process of the proteins once the mutated protein has been inoculated,» rejoices Marc Dhenain.
The next step will be to transmit this amyloid form to humans. This is a long-term project as no clinical trial can be considered for at least five years.
By Marine Laplace
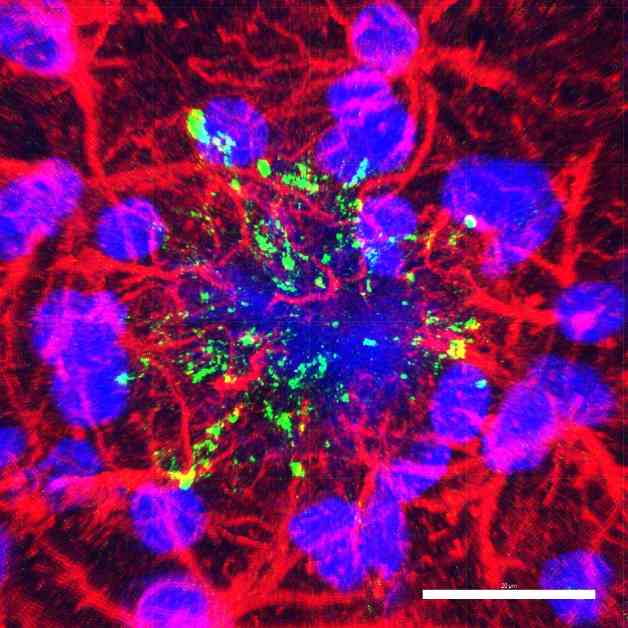
Opening image: Tau lesions of Alzheimer’s disease in green surrounding an amyloid lesion (large blue area). The red elements are brain cells. The other blue elements are cell nuclei.
Credits: Fanny PETIT